Introduction
Chemical Dominos was a project where each group created a board game. The goal was to find a fun way to witness and learn about chemicals and their reactions. Some of our focuses for this project were reading and writing chemical equations, single replacement reactions, double displacement reactions, creation of a gas, and dealing with LEDs and Breadboards. To do this, my group created a board game with a candy theme, titled Land of Candy. The game had players choose game pieces (various types of candy), and select cards to make their way to the finish line. Along the way some cards had special instructions that told participants ways to combine chemicals to create reactions. All of these reactions will be later explained, in addition to how we managed to intertwine them with our theme.
Instructions
For our project, I typed up, printed out, and laminated the Land of Candy document shown below for the audience to view. In addition to this, the instructions and description were part of our presentation. One group member read the Description and instructions at the beginning of the game.
Scripts
To create an easy flowing presentation that seemed rehearsed and professional, I typed out scripts for each of our group members. We all had a portion of the information that we needed to present at strategic times throughout our game. I printed out the completed script document, the cut out each person's individual script. From there I laminated them with a candy assorted backside to help make an aesthetically pleasing card for the audience to look at.
Final Project Photos
Blueprint
In addition, I made a blueprint of our game board. The blueprint had accurate dimensions, displaying all obstacles and component placement. This included our Hershey kiss border, makeshift lollipop, candy corn, and all spots for players to land on. We also included the few safety requirements our game had. The safety requirement list was not very extensive due to the relatively safe nature of our chemicals. This made the majority of our safety precautions common sense.
The Chemistry
One chemistry concept studied and discussed throughout the project was single replacement reactions. Our games specific reaction featured Aluminum and Copper Nitrate. A single replacement reaction, also known as a single reaction or a single-displacement reaction, is where an element reacts with a compound and replaces an element in that compound. This its often represented using A, B, and C blocks featured in the diagram below.
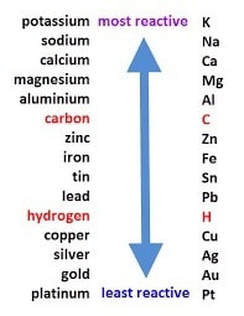
In this our game, Aluminum (Al(s)) and Copper Nitrate (Cu(NO3)(l)) React to form Copper (Cu(s)) and Aluminum Nitrate (Al(NO3)3. I chose this product specifically because it has a capability to dissolve certain substances. In our game, Copper and Aluminum Nitrate, the solution created, dissolves a piece of aluminum, which breaks a chain of electric current, lighting an LED. We know this works because of the reactivity series shown in the chart below.
The reactivity series is a list of metals listed in a specific order. The metals are ordered by their level of reactivity, platinum being the least reactive and potassium being the most. This works in our game because metal A, Aluminum, replaces metal B, Copper. Since Aluminum is higher on the reactivity series, the single displacement reaction occurs. We tied this reaction into our theme by having the solution and end product renamed. One of our card instructions told the player to add blue raspberry juice (Copper Nitrate) into a tin containing aluminum. The blue raspberry idea follows our candy theme. In addition to this, the LED the reaction lit was the nose of a reindeer, creating a festive holiday component of our game.
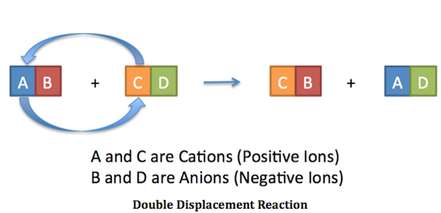
Another concept we studied for Land of Candy is double replacement reactions. A double displacement reaction, also known as a double replacement reaction or a metathesis reaction, is where the anions and cations switch between two reactants, forming new products. This reaction is often demonstrated using A, B, C, and D blocks shown in the diagram below.
Another concept we studied for Land of Candy is double replacement reactions. A double displacement reaction, also known as a double replacement reaction or a metathesis reaction, is where the anions and cations switch between two reactants, forming new products. This reaction is often demonstrated using A, B, C, and D blocks shown in the diagram below.
Our double displacement reaction features Sodium Acetate (C2H3NaO2(l)) and Iron(III) Chloride (FeCl3(l)). These two chemicals react to form Iron Acetate (Fe(C2H3O2)2(s)) and Sodium Chloride (NaCl(s)). We chose this combination because of the color it creates when Sodium Acetate and Iron(III) Chloride mix. The solution is a vibrant orange color that we integrated into our Land of Candy theme. We had the frame of a Candy Corn set up, with a clear solution (Sodium Acetate) where the orange should have been. One card had instructions to add Orange Soda (Iron(III) Chloride) to the clear solution, which will complete the candy corn.
Finally,we studied creations of gas. We did this by completing many mini labs to see how it worked, and how to write out these reactions. Our Land of Candy featured
a creation of gas by combining vinegar (HCH3COO(l)) and baking soda (NaHCO3(s)). When these two combine, carbon (CO2(g)), water (H2O(l)), and sodium acetate (NaC2H3O2(s)) are produced. This is an acid based reaction. Because this is the most visible and captivating reaction, we used this as our final reaction. This reaction's instructions were on the winning card, marking the end of our game. We had priorly set up vinegar in an up-right PVC pipe, with a balloon on top. That balloon sags to the side, weighted down by baking soda inside. To continue along with our theme, we asked the winning player to pour the "sugar" in the balloon down the pipe. We then referred to the inflated ballon atop the white pipe as a lollipop. All of this Chemical reaction information was displayed as a laminated document that I created and printed, placed at the base of our table for the audience to read.
Finally,we studied creations of gas. We did this by completing many mini labs to see how it worked, and how to write out these reactions. Our Land of Candy featured
a creation of gas by combining vinegar (HCH3COO(l)) and baking soda (NaHCO3(s)). When these two combine, carbon (CO2(g)), water (H2O(l)), and sodium acetate (NaC2H3O2(s)) are produced. This is an acid based reaction. Because this is the most visible and captivating reaction, we used this as our final reaction. This reaction's instructions were on the winning card, marking the end of our game. We had priorly set up vinegar in an up-right PVC pipe, with a balloon on top. That balloon sags to the side, weighted down by baking soda inside. To continue along with our theme, we asked the winning player to pour the "sugar" in the balloon down the pipe. We then referred to the inflated ballon atop the white pipe as a lollipop. All of this Chemical reaction information was displayed as a laminated document that I created and printed, placed at the base of our table for the audience to read.
Reflection
Overall, I feel that the project was a success. We came together and created a finished product that informed everyone about our chemical concepts, as well as entertained them though a game. As for me personally, I personally think I did well as the leader of the group. I think I was able to divide work between group members efficiently. There were certain tasks that I felt some group members could preform at a higher level than other, and so divvying up those tasks was one of my strong-suits. This helped to get tasks such as painting or hot gluing done quicker. Another peak of mine was the production of all of our documents. This peak would fall under the categories work ethic and productivity. I single handedly typed up our instructions, chemical reactions, scripts, and drew our blueprints with safety precautions. This peak made me realize how productive I could be when given a limited amount t of time and plenty of determination.
A weak-point of mine was empathy. Although this was not terrible by any means, I felt I could have been more empathetic towards people throughout the project. There were some instances where I could have said things nicer, or been more mindful of moods certain group members were in. This could have helped those group members be in better spirits, feeling more motivated to create a project they were proud of. Another pit of mine was organization. This particular project had many moving parts that were difficult to bring together for a final product. There were so many little components such as the placement of the wiring to the individual card instructions that had to correspond in such a way to create a good finished product. Unfortunately, I felt our final product reflects this all too much, and could have been put together more neatly. If I were to redo this product, I would try to focus more attention on creating a neat, organized looking game board.
A weak-point of mine was empathy. Although this was not terrible by any means, I felt I could have been more empathetic towards people throughout the project. There were some instances where I could have said things nicer, or been more mindful of moods certain group members were in. This could have helped those group members be in better spirits, feeling more motivated to create a project they were proud of. Another pit of mine was organization. This particular project had many moving parts that were difficult to bring together for a final product. There were so many little components such as the placement of the wiring to the individual card instructions that had to correspond in such a way to create a good finished product. Unfortunately, I felt our final product reflects this all too much, and could have been put together more neatly. If I were to redo this product, I would try to focus more attention on creating a neat, organized looking game board.
Summary
Overall, this was an extremely fun project. I personally got the chance to learn a great deal about different reactions, dealing with chemical equations, and electric currents, all while being able to teach it to others. This is a great interactive way to get students involved and I would recommend amend to any chemistry teacher given the opportunity.